The vaccine efficacy measure relevant for COVID-19 epidemiology, should be based on real-world data, not on clinical data which have data selection biases indicated in this paper. Recent United Kingdom data suggest that age-dependent asymptomatic breakthrough cases and natural immunity of the unvaccinated, may have led to negative vaccine efficacy where vaccinated infectivity is higher than unvaccinated infectivity among major age groups. The phenomenon of relative risk enhancement of vaccines among certain cohorts suggests diminishing effectiveness of the vaccination strategy for the COVID-19 pandemic.
Introduction
In March 2020, the World Health Organization (WHO) declared [1] the COVID-19 pandemic based on real-world data which showed a rapid rise in reported cases within a few weeks in several countries. Since then, public health policy has been focussed on containing the rates of infection, with the data being collected and published by national agencies such as the UK Health Security Agency (UKHSA) [2] among others. National data are collated into global databases such as Our World in Data (OWID) [3] which are the raw data free from the adjustment and selection of clinical studies. Epidemiologically, the real-world raw data provide the most relevant information because they drive policy decisions.
Recent research shows that, for some countries, the vaccinated population remains a significant source of COVID-19 transmission [4], and no evidence of short-term correlation was found [5] between COVID-19 cases and the level of vaccination in most countries. Furthermore, a statistically significant adverse correlation was found [6] over the long-term among 98 countries, indicating higher levels of vaccination are positively correlated with greater increases in COVID-19 infection. These observations of significant infection among the vaccinated raise doubts about vaccine efficacy for COVID-19 epidemiology.
Yet these real-world evidences of vaccine ineffectiveness in infection mitigation have been summarily dismissed due to the firmly held assumption that “vaccines are highly effective”. Globally, mass vaccination strategy for COVID-19 pandemic has been based on the encouraging results of original vaccine trials, such as that of Pfizer-BioNTech [7] and more recent clinical studies [8,9] which have maintained that COVID vaccines are effective in reducing the risk of infection. Yet results from real-world data cited above suggest vaccines are ineffective in reducing infection and transmission of COVID-19 disease.
Paradoxically, governments and health agencies such as the UKHSA [2] have deprecated and ignored their own data which drive public policy and instead held firm to the “highly effective” claims on vaccine efficacy by the vaccine manufacturers. The disconnect between positive vaccine effectiveness shown in clinical data and negative vaccine effectiveness observed in real-world data, urgently needs resolution, because the inconsistency between official assumptions and observed outcomes is scientifically intolerable. This paper resolves the contradiction by showing different measures of vaccine efficacy are appropriate for measuring effectiveness on different populations. With this new understanding, UKHSA data are used to estimate vaccine efficacy in COVID-19 epidemiology for the United Kingdom.
Data Sources
Vaccine efficacy estimates depend on sampling of vaccinated and unvaccinated populations. In COVID vaccines trials, such as that of Pfizer-BioNTech [7], and in most clinical studies [8,9], past infected persons are ruled out of participation [7] because “key exclusion criteria included a medical history of Covid-19”. The method applied in hospitals and medical clinics is typically the “test-negative case–control design” (TNCCD) [10]. The samples are biased to include mostly patients or users of the medical facilities. Indeed, the studies [8] explicitly stated that their experimental design was “to estimate the effectiveness of vaccination against symptomatic disease”. That is, infection-recovered persons and asymptomatic cases are excluded.
The TNCCD approach may be inappropriate for COVID-19 epidemiology because it includes only a subset of the population, excluding infection-recovered and asymptomatic participants. Asymptomatic cases are those who tested positive, while showing no symptoms. The vaccine efficacy estimated by the vaccine manufacturers may be accurate and useful for symptomatic individuals who have not previously been infected with COVID-19. However, this subset is not sufficiently representative of the whole population because the missing cohorts in the subset constitute significant percentages of the population. A recent review [11] covering nearly 30 million individuals reported that over 40% of confirmed COVID-19 infections are asymptomatic. There are also substantial and increasing percentages of populations with natural immunity acquired from past infections. From data on US blood donation [12], over 20% of the general US population was found in May 2021 to have infection-induced immunity and in August 2021, CDC observed [13] about half of infected adults (with antibodies) were unvaccinated.
The UKHSA [2] has warned that “comparing case rates among vaccinated and unvaccinated populations should not be used to estimate vaccine effectiveness against COVID-19 infection”, because it has assumed as correct the vaccine effectiveness estimated by the vaccine manufacturers, which differ substantially from the vaccine effectiveness calculated from its own data, as will be seen below. On the contrary, vaccine efficacy estimated from the UKHSA data is most relevant and pertinent for public health policy on COVID-19 epidemiology in the United Kingdom.
Methods
The data used in this paper for COVID-19 epidemiology in the United Kingdom are assembled from the UKHSA weekly COVID-19 surveillance reports [2]: Table 5 from Week 43 and 44 reports; Table 6 from Week 45 and 46; Table 11 from Week 47 to 51 reports. The datasets cover the period from 27 September to 17 December 2021. The data used are unadjusted rates of infection per 100,000 based on cases reported by specimen data, where persons vaccinated with two doses are compared to those unvaccinated to assess vaccine efficacy.
Vaccine efficacy or effectiveness in preventing infection is usually measured by relative risk reduction (RRR), even though absolute risk reduction (ARR) conveys a more accurate and informative measure of benefit for the vaccine recipient. The UKHSA data [2] are used firstly to calculate vaccine ARR which is defined as unvaccinated infectivity minus vaccinated infectivity, where infectivity is defined by reported cases of the group per 100,000 population, then RRR is calculated as the ratio of ARR to unvaccinated infectivity.
Findings
At this writing, the latest of UKHSA surveillance report (Week 51, published 23 December) has an example of the relevant data in Table 11, on page 40. The two data fields for vaccinated and unvaccinated infectivity are cases reported over the previous four-week period, expressed as unadjusted rates per 100,000, among persons vaccinated with two doses and among persons not vaccinated. These two data fields found in different locations of earlier surveillance reports are assembled and used to calculate vaccine efficacy for different age groups either over rolling four-week periods or over three discrete four-week periods. The latter results are shown in Figure 1.
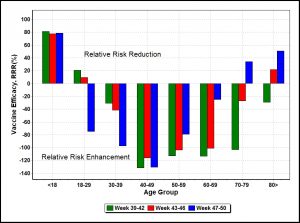 Figure 1. Vaccine efficacy measured by relative risk reduction versus age groups in United Kingdom (Week 39-50)
Figure 1. Vaccine efficacy measured by relative risk reduction versus age groups in United Kingdom (Week 39-50)
The most striking impression is the presence of significant negative vaccine efficacy. The UKHSA [2] expects from clinical data that relative risk reduction (RRR) is positive with a range 65% to 95% around the 80% level shown for the under-18 age group in Figure 1. However, from unadjusted UKHSA data, vaccine efficacy for all other age groups, falls well short of expectation, though the under 18 age group and the over 70s still mostly show infection relative risk reduction. Examination of relevant UKHSA tables shows that all age groups have significant numbers of reported vaccinated cases, called breakthrough cases. In the main 30-70 age groups breakthrough cases substantially exceed unvaccinated cases.
Note that vaccine efficacy changes over time due to changing circumstances, including behavioural changes, for example, in the over 80s age group, vaccines improved over a 12-week period from being ineffective to being effective. The most recent period saw a significant deterioration of vaccine efficacy in the socially active 18-40 age groups, perhaps due to more Omicron testing incentivized by holiday overseas travel, whereas the over 70s age groups saw improvements in vaccine efficacy.
Evidently in Figure 1, for many age groups between 30-70. data bars point downwards, indicating those cohorts generally have vaccinated infectivity significantly higher than unvaccinated infectivity, leading to deeply negative ARR and RRR or negative vaccine efficacy. Instead of relative risk reduction, vaccines were associated with relative risk enhancement (RRE) for the bulk of the working population. In the significant 40-70 age group, vaccines could double the risk of infection relative to the unvaccinated.
In summary, vaccine efficacy was highly variable over time and strongly dependent on age, with the working 30-70 age groups showing substantial relative risk enhancement from vaccination. Though this observation is limited to only a 12-week period and it may not be permanent, nevertheless it falsifies the widely-held assumption that vaccination is always beneficial for everyone in the COVID-19 pandemic.
Interpretation
UKHSA [2] has cautioned the data are unadjusted for potential statistical biases which may arise from systematic differences between the vaccinated and unvaccinated populations. The examples given include: the vaccinated are vaccinated because they may be more health conscious, but also may want greater levels of social interaction, while the unvaccinated may have recovered from previous infections. Not mentioned are some unvaccinated individuals who may be highly health conscious using prophylaxis and therapies which may be more effective than commonly thought. Adjusting for these factors, if possible, may be relevant for informing a previously uninfected individual on how the vaccines may affect their symptomatic infection risk, but it is inappropriate for epidemiology which has the objective of assessing vaccine performance in a complex real-world situation including all persons.
Highly adjusted and controlled clinical data may be appropriate under fixed and homogeneous clinical conditions. Those conditions clearly do not reflect the real-world which is both dynamic and heterogeneous. Governments and health authorities have made, and continuously maintained, the fixed policy assumption that vaccines are highly effective for everyone always, to justify mass vaccination. This assumption has been falsified by current findings and by other studies mentioned above [4-6]. A simple reason for why fixed assumptions are not justified is that the vaccines themselves may have caused [14] rapid and significant changes to the epidemiology.
The UKHSA [15] has put under surveillance over 20 SARS-CoV-2 variants, of which five are variants of concern (VOC), six are variants under investigation (VUI) and at least 12 are variants in monitoring (VIM). Eleven have Greek alphabet names, of which Delta and Omicron are currently the most dominant variants for the COVID-19 pandemic. Clearly, new variants may render vaccines which were developed against previous variants less effective, because the selection pressure on viral mutation is specifically to overcome the defence of previously vaccine-induced antibodies.
The rapid appearance of new COVID variants may be caused [14] by vaccine-mediated immune escape, which is consistent with the above findings of relative risk enhancement of the vaccinated. In the United Kingdom, the new Omicron variant probably emerged in early November and was detected two weeks later. Figure 1 shows (blue bars) the Omicron variant appears to affect more severely the vaccinated younger age groups than in the unvaccinated or the older age groups.
Two cohorts ignored by clinical data, as mentioned above: asymptomatic and infection-recovered population with natural immunity are expected to continue to increase, because that growing community is integral to the process of developing herd immunity. There is also mounting evidence [16] that natural immunity is superior to vaccine-induced immunity against fast mutating coronaviruses. The burgeoning numbers of those who have acquired herd immunity may render COVID-19 endemic like the common cold, making vaccination relatively less relevant in future.
Conclusion
Vaccine efficacy for COVID epidemiology should be measured using real-world data and not clinical data which have significant data selection bias. United Kingdom data [2] show vaccine efficacy is dynamic and age-dependent, with vaccine relative risk enhancement occurring significantly in some age groups. Both more asymptomatic breakthrough cases and more infection-recovered cases among the unvaccinated may explained why vaccines have recently had negative efficacy.
The unwavering, one-size-fits-all policy of mass vaccination, disregarding changing and accumulating contrary evidence may be exacerbating the COVID-19 pandemic. Policy initiatives and problem perception are based on real-world data, whereas the vaccine solution is inconsistently based on clinical data. As the pandemic advances, with herd immunity spreading, vaccines have become relatively ineffective, but unrecognized by clinical data, due to new variants likely induced by vaccine-mediated immune escape [14,15] with effective natural immunity of the unvaccinated [16] in major age groups downplayed or ignored.
Reference
- Ghebreyesus TA. WHO Director-General's opening remarks at the media briefing on COVID-19 - 11 March 2020. Retrieved from: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed Dec 20, 2021).
- UK Health Security Agency. COVID-19 vaccine surveillance reports (weeks 39 to 51), Retrieved from: https://www.gov.uk/government/publications/covid-19-vaccine-weekly-surveillance-reports (accessed Dec 27, 2021).
- Ritchie H, Mathieu E, Rodés-Guirao L et al. Coronavirus pandemic (COVID-19). 2020. Published online at OurWorldInData.org. Retrieved from: https://ourworldindata.org/coronavirus (accessed Nov 5, 2021).
- Kampf G. The epidemiological relevance of the COVID-19-vaccinated population is increasing, The Lancet Regional Health – Europe 11 (Dec. 2021) 100272. https://www.sciencedirect.com/science/article/pii/S2666776221002581?via%3Dih (accessed Dec 12, 2021).
- Subramanian SV, Kumar A. Increases in COVID‑19 are unrelated to levels of vaccination across 68 countries and 2947 counties in the United States, European Journal of Epidemiology. https://link.springer.com/content/pdf/10.1007/s10654-021-00808-7.pdf (accessed Dec 12, 2021).
- Sy W. Epidemiological impact of COVID-19 fully vaccinated population. Retrieved from: https://principia-scientific.com/epidemiological-impact-of-covid-19-fully-vaccinated-population/ (accessed Jan 27, 2022).
- Polack FP, Thomas SJ et al. Safety and efficacy of BNT162b2 mRNA Covid-19 vaccine, The New England Journal of Medicine, December 16, 2020, Vol. 383, No.27. Retrieved from: https://www.nejm.org/doi/pdf/10.1056/NEJMoa2034577?articleTools=true (accessed Dec 16, 2021).
- Bernal JL, Andrews N et al. Effectiveness of Covid-19 Vaccines against the B.1.617.2
(Delta) Variant, The New England Journal of Medicine, August 12, 2021, Vol. 385, No.7. Retrieved from: https://www.nejm.org/doi/pdf/10.1056/NEJMoa2108891?articleTools=true (accessed Dec 19, 2021). - Andrews N, Tessier E et al. Vaccine effectiveness and duration of protection of Comirnaty, Vaxzevria and Spikevax against mild and severe COVID-19 in the UK. medRxiv. October 6, 2021. Retrieved from: https://www.medrxiv.org/content/10.1101/2021.09.15.21263583v2.full.pdf (accessed Dec 19, 2021)
- Vandenbroucke JP, Pearce, N. Test-Negative Designs: Differences and Commonalities with Other Case-Control Studies with "Other Patient" Controls, Epidemiology, November 30, 2019, Vol. 6, 838-844. Retrieved from: https://pubmed.ncbi.nlm.nih.gov/31430265/ (accessed Dec 19, 2021).
- Ma Q, Liu J et al. Global percentage of asymptomatic SARS-CoV-2 infections among the tested population and individuals with confirmed COVID-19 diagnosis: a systematic review and meta-analysis, JAMA Network Open, December 14, 2021, Vol. 4, No. 12, e2137257. Retrieved from: https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2787098?utm_source=silverchair&utm_medium=email&utm_campaign=article_alert-jamanetworkopen&utm_content=wklyforyou&utm_term=121521 (accessed Dec 23, 2021).
- Jones JM, Stone, M et al. Estimated US infection- and vaccine-induced SARS-CoV-2 seroprevalence based on blood donations, July 2020-May 2021, JAMA. September 2, 2021; Vol. 326, No. 14:1400-1409. Retrieved from: https://jamanetwork.com/journals/jama/fullarticle/2784013 (accessed Dec 24, 2021).
- Centers for Disease Control and Prevention (U.S.). Science brief: SARS-CoV-2 infection-induced and vaccine-induced immunity, Updated October 29, 2021. Retrieved from: https://www.cdc.gov/coronavirus/2019-ncov/science/science-briefs/vaccine-induced-immunity.html (accessed Dec 24, 2021).
- Harvey WT, Carabelli AM, Jackson B et al. SARS-CoV-2 variants, spike mutations and immune escape, Nature Reviews Microbiology, 2021 June 1: 1-18. Retrieved from: https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC8167834/pdf/41579_2021_Article_573.pdf (accessed Dec 12, 2021).
- UK Health Security Agency. SARS-CoV-2 variants of concern and variants under investigation in England, Technical briefing 33, December 23, 2021, Retrieved from: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1043807/technical-briefing-33.pdf (accessed Dec 27, 2021).
- Gazit S, Shlezinger R et al. Comparing SARS-CoV-2 natural immunity to vaccine-induced immunity: reinfections versus breakthrough infections, medRxiv. August 25, 2021. Retrieved from: https://www.medrxiv.org/content/10.1101/2021.08.24.21262415v1.full.pdf (accessed Dec 27, 2021).
